This web page was produced as an assignment for Genetics 564, an undergraduate course at UW-Madison
the tub protein
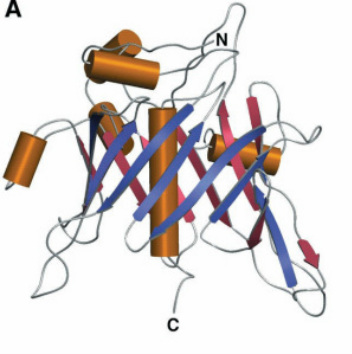 Figure 1. The TUB protein 3-D structure
(from Boggon 1999 Figure 2A).
Figure 1. The TUB protein 3-D structure
(from Boggon 1999 Figure 2A).
Two major isoforms of the TUB protein (NP_003311.2) normally exist due to the two splice variants.These isoforms only differ at their far N-terminus and do not appear to have any difference in function or location. The 561 amino acid, isoform a, has been used throughout this site as the TUB protein. TUB proteins are normally found in the same locations as the mRNA, namely the nucleolus of paraventricular (PVN), ventromedial (VMH), and arcuate nuclei of the hypothalamus as well as the retina and cochlea. [1]
While the TUB protein is largely hydrophilic by nature, the conserved TUB domain at its C-terminal end contains a positively-charged, hydrophobic core that binds to double-stranded DNA, pointing to its function as a transcription factor within the hypothalamus (Figure 1). [2] This TUB domain (shown in Figure 2) has also been shown to interact with Gαq proteins at the plasma membrane leading to its partial localization to the plasma membrane. Upon Gαq protein coupled receptor activation, TUB relocates to the nucleus where it could interact with DNA to modulate gene expression. The nuclear localization of TUB is due to a nuclear localization signal (K39KKR) in its N-terminal domain (Figure 3). [3] While the TUB domain is highly conserved all the way back to plants, the N-terminal portion of the protein, and its nuclear localization signal is not well conserved in length or sequence (Figure 2).
While the TUB protein is largely hydrophilic by nature, the conserved TUB domain at its C-terminal end contains a positively-charged, hydrophobic core that binds to double-stranded DNA, pointing to its function as a transcription factor within the hypothalamus (Figure 1). [2] This TUB domain (shown in Figure 2) has also been shown to interact with Gαq proteins at the plasma membrane leading to its partial localization to the plasma membrane. Upon Gαq protein coupled receptor activation, TUB relocates to the nucleus where it could interact with DNA to modulate gene expression. The nuclear localization of TUB is due to a nuclear localization signal (K39KKR) in its N-terminal domain (Figure 3). [3] While the TUB domain is highly conserved all the way back to plants, the N-terminal portion of the protein, and its nuclear localization signal is not well conserved in length or sequence (Figure 2).
TUBBY Mouse mutation
The splice site transversion that spontaneously arose in tubby mice results in the last 44 amino acids of the carboxyl terminus being replaced by 20 different amino acids and early truncation of the protein within the highly conserved TUB domain. [4] This disruption of the C-terminus disrupts the protein's ability to bind to DNA or G-proteins at the plasma membrane. This truncation also appears to affect the stability of the protein itself, as levels of the TUB protein are nearly undetectable in tubby mice. [5] Interestingly, TUB expression is unaffected in other obesity models (ob, db, or melanocortin 4 receptor-deficient mice) indicating that the TUB protein may function in a novel pathway to modulate energy homeostasis. [5]
HYPOTHESES about the TUB protein pathway
TUB AFFECTS FEEDING BEHAVIOR
The localization of TUB to the hypothalamus indicates that it may be involved in regulation of feeding behavior. This was confirmed as tubby mice have a higher food consumption as compared to wild-type littermates. [6] The anorexigenic neuropeptide POMC, which normally reduces food intake is found at lower levels in tubby mice, consistent with their overfeeding. [7] Similarly, the orexigenic neuropeptide, orexin, which normally increases food intake is found at higher levels in tubby mice. [8] These results indicate that TUB may normally regulate the expression of these neuropeptides. Tubby mice also fail to activate their carbohydrate metabolism leading to a reliance on fat metabolism for energy needs. [8] Similarly, human polymorphisms in the Tub gene were found to be correlated with increased carbohydrate intake in women pointing to TUB's function in regulating eating behavior in mice as well as humans. [9] This hypothesis is further explored in my specific aims.
TUB IS AN INSULIN, LEPTIN, OR SEROTONIN SUBSTRATE
The TUB protein is phosphorylated by both the insulin receptor (IRTK) in response to insulin [10] leptin receptor (LEPR) JAK2 in response to leptin [11] leading to its translocation to the nucleus [11,12] . This indicates that TUB may be a direct substrate of insulin and leptin. This is supported by the fact that mice treated with anti-sense TUB lose the ability to respond to insulin and leptin, resulting in a loss of food intake regulation normally provided by these signals. [11] Stimulation of the serotonin receptor 5HT2C also results in translocation of TUB into the nucleus, and mice with mutated 5HT2C display a phenotype very similar to tubby mice indicating that TUB may also function downstream of serotonin signaling. [3]
TUB SERVES AS A PHAGOCYTOSIS SIGNAL
In the retina, TUB has been shown to provide "eat me" signals by serving as a substrate of the MerTK ligand to facilitate phagocytosis. [13] In a similar fashion, TUB has also been shown to be secreted from cells in a pathway not mediated by the endoplasmic reticulum-Golgi apparatus pathway as a signal to macrophages to phagocytose. [14] It is still unclear, however, whether phagocytosis may be involved in the pathway leading to the tubby obesity phenotype.
The localization of TUB to the hypothalamus indicates that it may be involved in regulation of feeding behavior. This was confirmed as tubby mice have a higher food consumption as compared to wild-type littermates. [6] The anorexigenic neuropeptide POMC, which normally reduces food intake is found at lower levels in tubby mice, consistent with their overfeeding. [7] Similarly, the orexigenic neuropeptide, orexin, which normally increases food intake is found at higher levels in tubby mice. [8] These results indicate that TUB may normally regulate the expression of these neuropeptides. Tubby mice also fail to activate their carbohydrate metabolism leading to a reliance on fat metabolism for energy needs. [8] Similarly, human polymorphisms in the Tub gene were found to be correlated with increased carbohydrate intake in women pointing to TUB's function in regulating eating behavior in mice as well as humans. [9] This hypothesis is further explored in my specific aims.
TUB IS AN INSULIN, LEPTIN, OR SEROTONIN SUBSTRATE
The TUB protein is phosphorylated by both the insulin receptor (IRTK) in response to insulin [10] leptin receptor (LEPR) JAK2 in response to leptin [11] leading to its translocation to the nucleus [11,12] . This indicates that TUB may be a direct substrate of insulin and leptin. This is supported by the fact that mice treated with anti-sense TUB lose the ability to respond to insulin and leptin, resulting in a loss of food intake regulation normally provided by these signals. [11] Stimulation of the serotonin receptor 5HT2C also results in translocation of TUB into the nucleus, and mice with mutated 5HT2C display a phenotype very similar to tubby mice indicating that TUB may also function downstream of serotonin signaling. [3]
TUB SERVES AS A PHAGOCYTOSIS SIGNAL
In the retina, TUB has been shown to provide "eat me" signals by serving as a substrate of the MerTK ligand to facilitate phagocytosis. [13] In a similar fashion, TUB has also been shown to be secreted from cells in a pathway not mediated by the endoplasmic reticulum-Golgi apparatus pathway as a signal to macrophages to phagocytose. [14] It is still unclear, however, whether phagocytosis may be involved in the pathway leading to the tubby obesity phenotype.
references
Cover Photo Credit
[1] He, W., Ikeda, S., Bronson, R.T., Yan, G., Nishina, P.M., North, M.A., and Naggert, J.K. (2000). GFP-tagged expression and immunohistochemical studies to determine the subcellular localization of the tubby gene family members. Molecular Brain Research, 81(1), 109. doi:10.1016/S0169-328X(00)00164-9.
[2] Boggon, T.J., Shan. W.S., Santagata, S., Myers, S.C., & Shapiro, L. (1999). Implication of tubby proteins as transcription factors by structure-based functional analysis. Science, 286(5447) doi: 10.1126/science.286.5447.2119
[3] Santagata, S., Boggon, T.J., Baird, C.L., Gomex, C.A., Zhao, J., Shan, S., Myszka, D.G., & Shapiro, L. (2001). G-Protein Signaling Through Tubby Proteins. Science, 292(5524).
[4] Kleyn, P.W., Fan, W., Kovats, S. G., et al. (1996). Identification and Characterization of the Mouse Obesity Gene tubby: A Member of a Novel Gene Family. Cell, 85(2), doi:10.1016/S0092-8674(00)81104-6.
[5] Stubdal, H., Lynch, C.A., Moriarty, A., et al. (2000). Targeted Deletion of the tub Mouse Obesity Gene Reveals that tubby is a Loss-of-Function Mutation. Molecular and Cellular Biology, 20(3).
[6] Backberg, M., Madjid, N., Ogren, S.O., and Meister, B. (2004). Down-regulated expression of agouti-related protein (AGRP) mRNA in the hypothalamic arcuate nucleus of hyperphagic and obese tub/tub mice. Molecular Brain Research, 125 (129). doi:10.1016/j.molbrainres.2004.03.012.
[7] Guan, X.M., Yu, H., Lex, H.T., & Ploeg, V. (1998). Evidence of altered hypothalamic pro-opiomelancortin/neuropeptide Y mRNA expression in tubby mice. Molecular Brain Research, 59(273). doi: 10.1016/S0169328X98001508.
[8] Wang, Y., Seburn, K., Bechtel, L., Lee, B.Y., Szatkiewicz, J.P., Nishina, P.M., and Naggert, J.K. (2006). Defective carbohydrate metabolism in mice homozygous for the tubby mutation. Physiol Genomics, 27(131). doi:10.1152/physiolgenomics.00239.2005.
[9] Vliet-Ostaptchouk, J.V., Onland-Moret, N.C., Shiri-Sverdlov, R., et al. (2008). Polymorphisms of the TUB Gene Are Associated with Body Composition and Eating Behavior in Middle-Aged Women. PLoS ONE, 3(1), e1405. doi:10.1371/journal.pone.0001405.
[10] Kapeller, R., Moriarty, A., Strauss, A., Stubdal, H., Theriault, K., Siebert, E., Chickering, T., Morgenstern, J.P., Tartaglia, L.A., & Lillie, J. (1999). Tyrosine Phosphorylation of Tub and Its Association with Src Homology 2 Domain-containing Proteins Implicate Tub in Intracellular Signaling by Insulin. The Journal of Biological Chemistry, 274(35), 24980, doi:10.1074/jbc.274.35.24980.
[11] Prada, P.O., Quaresma, P.G.F., Caricilli, A.M., et al. (2013). Tub Has a Key Role in Insulin and Leptin Signaling and Action in Vivo in Hypothalamic Nuclei. Diabetes, 62(1),137, doi: 10.2337/db11-1388.
[12] Stretton, C., Litherland, G.J., Moynihan, A., Hajduch, E., & Hundal, H.S. (2009). Expression and modulation of TUB by insulin and thyroid hormone in primary rat and murine 3T3-L1 adipocytes, Biochemical and Biophysical Research Communications, 390(4), 1328, doi: 10.1016/j.bbrc.2009.10.147.
[13] Caberoy, N.B, Zhou, Y., & Li, W. (2010). Tubby and tubby-like protein 1 are new MerTK ligands for phagocytosis. The EMBO Journal, 29(3898). doi: 10.1038/emboj.2010.265.
[14] Caberoy, N.B. & Li, W. (2009). Unconventional secretion of tubby and tubby-like protein 1. FEBS Letters, 583(3057). doi:10.1016/j.febslet.2009.08.015.
[1] He, W., Ikeda, S., Bronson, R.T., Yan, G., Nishina, P.M., North, M.A., and Naggert, J.K. (2000). GFP-tagged expression and immunohistochemical studies to determine the subcellular localization of the tubby gene family members. Molecular Brain Research, 81(1), 109. doi:10.1016/S0169-328X(00)00164-9.
[2] Boggon, T.J., Shan. W.S., Santagata, S., Myers, S.C., & Shapiro, L. (1999). Implication of tubby proteins as transcription factors by structure-based functional analysis. Science, 286(5447) doi: 10.1126/science.286.5447.2119
[3] Santagata, S., Boggon, T.J., Baird, C.L., Gomex, C.A., Zhao, J., Shan, S., Myszka, D.G., & Shapiro, L. (2001). G-Protein Signaling Through Tubby Proteins. Science, 292(5524).
[4] Kleyn, P.W., Fan, W., Kovats, S. G., et al. (1996). Identification and Characterization of the Mouse Obesity Gene tubby: A Member of a Novel Gene Family. Cell, 85(2), doi:10.1016/S0092-8674(00)81104-6.
[5] Stubdal, H., Lynch, C.A., Moriarty, A., et al. (2000). Targeted Deletion of the tub Mouse Obesity Gene Reveals that tubby is a Loss-of-Function Mutation. Molecular and Cellular Biology, 20(3).
[6] Backberg, M., Madjid, N., Ogren, S.O., and Meister, B. (2004). Down-regulated expression of agouti-related protein (AGRP) mRNA in the hypothalamic arcuate nucleus of hyperphagic and obese tub/tub mice. Molecular Brain Research, 125 (129). doi:10.1016/j.molbrainres.2004.03.012.
[7] Guan, X.M., Yu, H., Lex, H.T., & Ploeg, V. (1998). Evidence of altered hypothalamic pro-opiomelancortin/neuropeptide Y mRNA expression in tubby mice. Molecular Brain Research, 59(273). doi: 10.1016/S0169328X98001508.
[8] Wang, Y., Seburn, K., Bechtel, L., Lee, B.Y., Szatkiewicz, J.P., Nishina, P.M., and Naggert, J.K. (2006). Defective carbohydrate metabolism in mice homozygous for the tubby mutation. Physiol Genomics, 27(131). doi:10.1152/physiolgenomics.00239.2005.
[9] Vliet-Ostaptchouk, J.V., Onland-Moret, N.C., Shiri-Sverdlov, R., et al. (2008). Polymorphisms of the TUB Gene Are Associated with Body Composition and Eating Behavior in Middle-Aged Women. PLoS ONE, 3(1), e1405. doi:10.1371/journal.pone.0001405.
[10] Kapeller, R., Moriarty, A., Strauss, A., Stubdal, H., Theriault, K., Siebert, E., Chickering, T., Morgenstern, J.P., Tartaglia, L.A., & Lillie, J. (1999). Tyrosine Phosphorylation of Tub and Its Association with Src Homology 2 Domain-containing Proteins Implicate Tub in Intracellular Signaling by Insulin. The Journal of Biological Chemistry, 274(35), 24980, doi:10.1074/jbc.274.35.24980.
[11] Prada, P.O., Quaresma, P.G.F., Caricilli, A.M., et al. (2013). Tub Has a Key Role in Insulin and Leptin Signaling and Action in Vivo in Hypothalamic Nuclei. Diabetes, 62(1),137, doi: 10.2337/db11-1388.
[12] Stretton, C., Litherland, G.J., Moynihan, A., Hajduch, E., & Hundal, H.S. (2009). Expression and modulation of TUB by insulin and thyroid hormone in primary rat and murine 3T3-L1 adipocytes, Biochemical and Biophysical Research Communications, 390(4), 1328, doi: 10.1016/j.bbrc.2009.10.147.
[13] Caberoy, N.B, Zhou, Y., & Li, W. (2010). Tubby and tubby-like protein 1 are new MerTK ligands for phagocytosis. The EMBO Journal, 29(3898). doi: 10.1038/emboj.2010.265.
[14] Caberoy, N.B. & Li, W. (2009). Unconventional secretion of tubby and tubby-like protein 1. FEBS Letters, 583(3057). doi:10.1016/j.febslet.2009.08.015.
Site created by Rachael Baird.
Genetics 564 Assignment, Spring 2014
University of Wisconsin-Madison
Last Updated: 5-10-14
Genetics 564 Assignment, Spring 2014
University of Wisconsin-Madison
Last Updated: 5-10-14


